[最も欲しかった] heterogeneous mixture of two elements 608429-Heterogeneous mixture of two elements
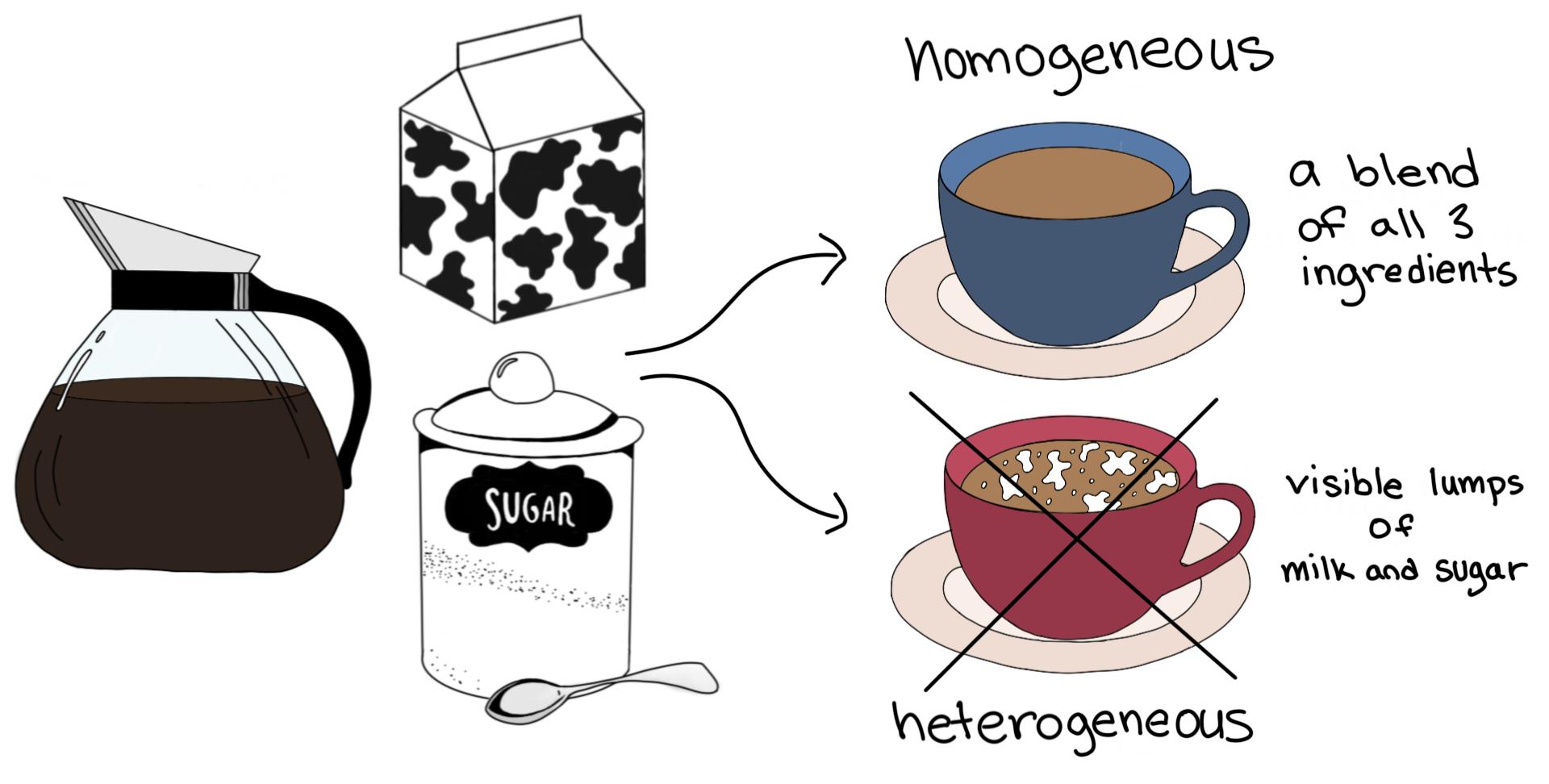
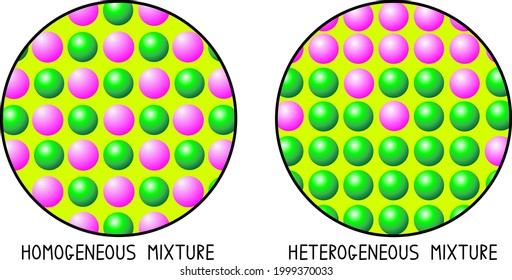
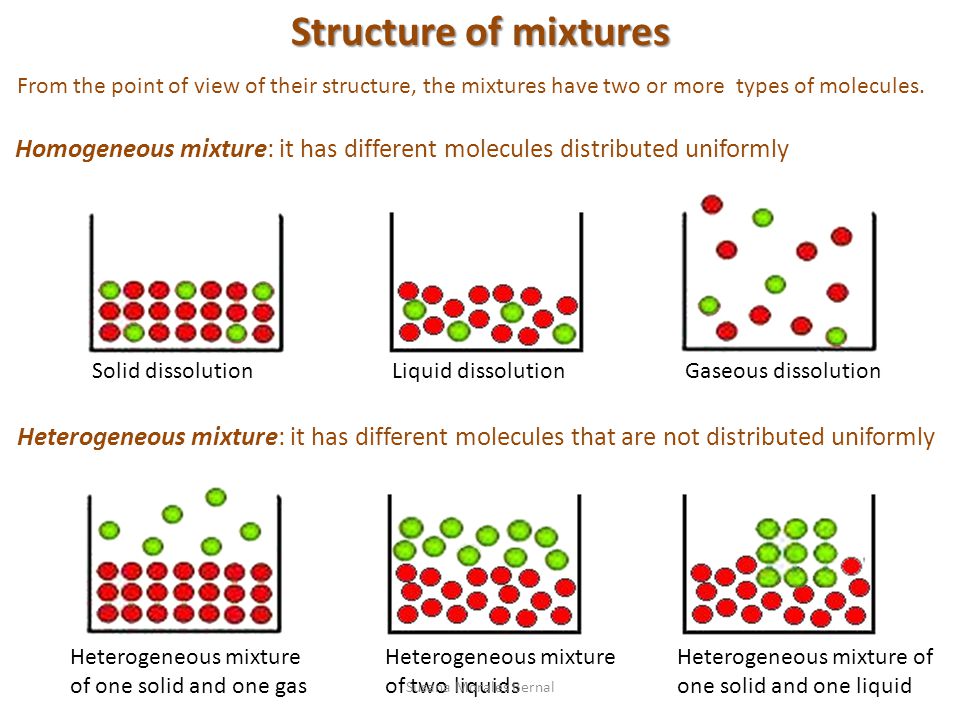
Heterogeneous Mixtures are those in which participants or components can be distinguished easily their composition is nonuniform also having their phase integrated irregularly and unevenly therefore it is possible to distinguish their phases with relative easeA MIXTURE is a combination of two or more substances that are not chemically united and do not exist in fixed proportions to each other Most natural substances are mixtures In the graphic on the left there are four substances water, alcohol, oil, and food color dyeA compound made up of two elements colloid a heterogeneous mixture with small particles dispersed in another medium that are small enough to stay suspended but large enough to scatter light compound two or more elements that have chemically combined dissolve

2 Elements P3 Sukwon Sci
Heterogeneous mixture of two elements
Heterogeneous mixture of two elements- Eg wine, beer, gelatin, water and alcohol Heterogeneous Mixtures Unlike homogeneous mixtures, in these it is very easy to identify, even to the naked eye, which are the different components that make them up This makes it much easier to separate these mixtures Eg water and oil, water and sand Most of the mixtures occurring in nature are heterogeneous For example, the soil is a mixture of hundreds of elements and compounds Its composition changes from place to place Some other examples of the heterogeneous mixture are – rocks, a mixture of kerosene and water, rice and pulses, etc




Elements Compounds And Mixtures Worksheet Chemistryrack
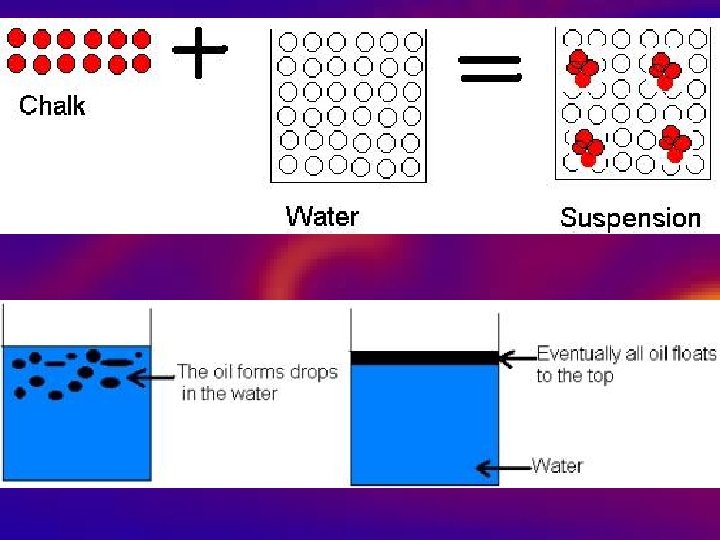
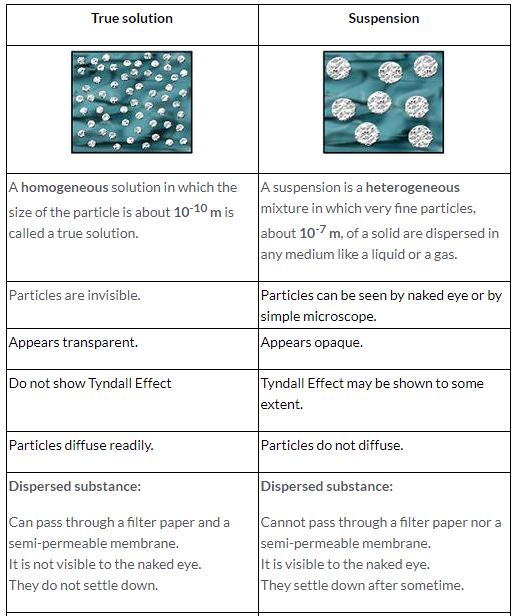
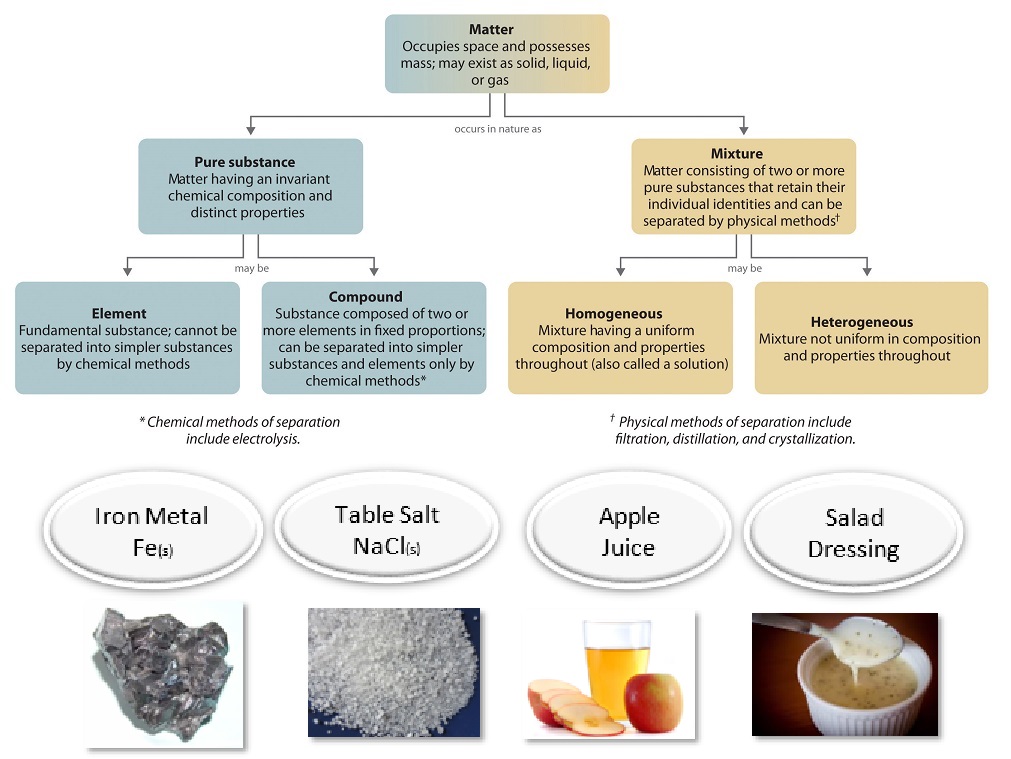
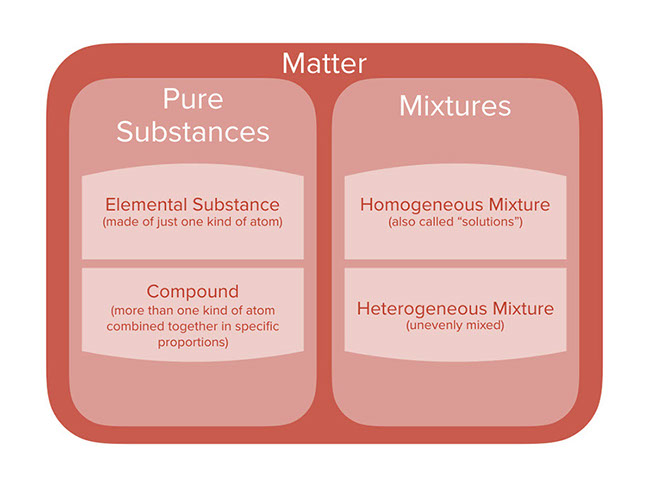
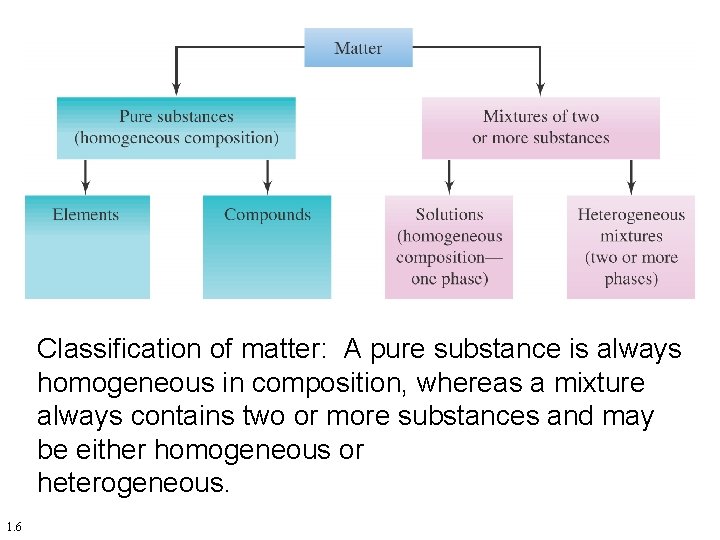
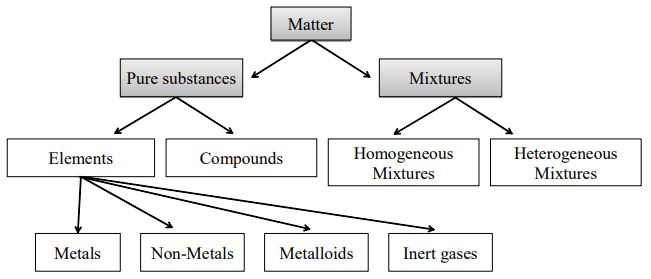
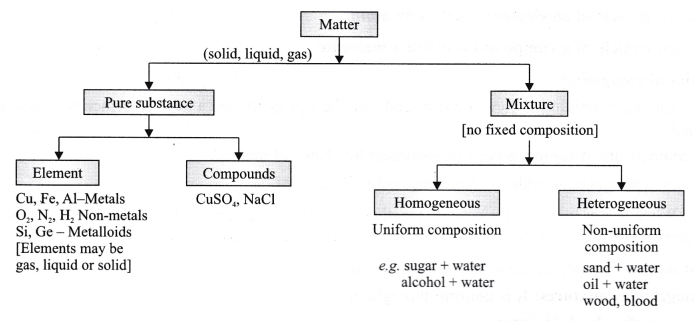
By definition, a pure substance or a homogeneous mixture consists of a single phase A heterogeneous mixture consists of two or more phases When oil and water are combined, they do not mix evenly, but instead form two separate layers Each of the layers is called a phaseYes, it is a type of homogeneous mixture It is because it hasA mixture may be either homogeneous or heterogeneous Mixtures may be composed of two or more elements, two or more compounds, or a combination of both Matter may be a pure substance or it may be a mixture A pure substance may either be an element or a compound All of these statements are true
The rainy atmosphere is a mixture of the air, which is a gas, with liquid rain droplets There are patches of dense liquid water falling down through the less dense air It's an obviously heterogeneous mixture because there are two states, or phases of matter liquid and gas Importantly, on a rainy day, visibility is limitedWhen two or more elements or compounds mix together, not necessarily in a definite ratio and do not interact chemically, then the resulting substance is know A mixture is a physical blend of two or more components, each of which retains its own identity and properties Only the form of the salt is changed when it is dissolved into water It retains its composition and chemical properties Homogeneous Mixtures A homogeneous mixture is a mixture in which the composition is uniform throughout the
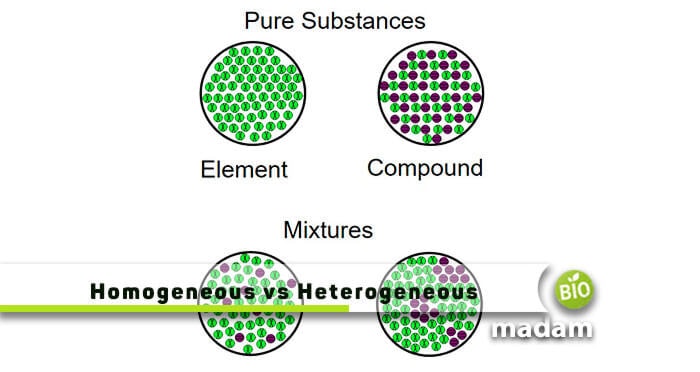
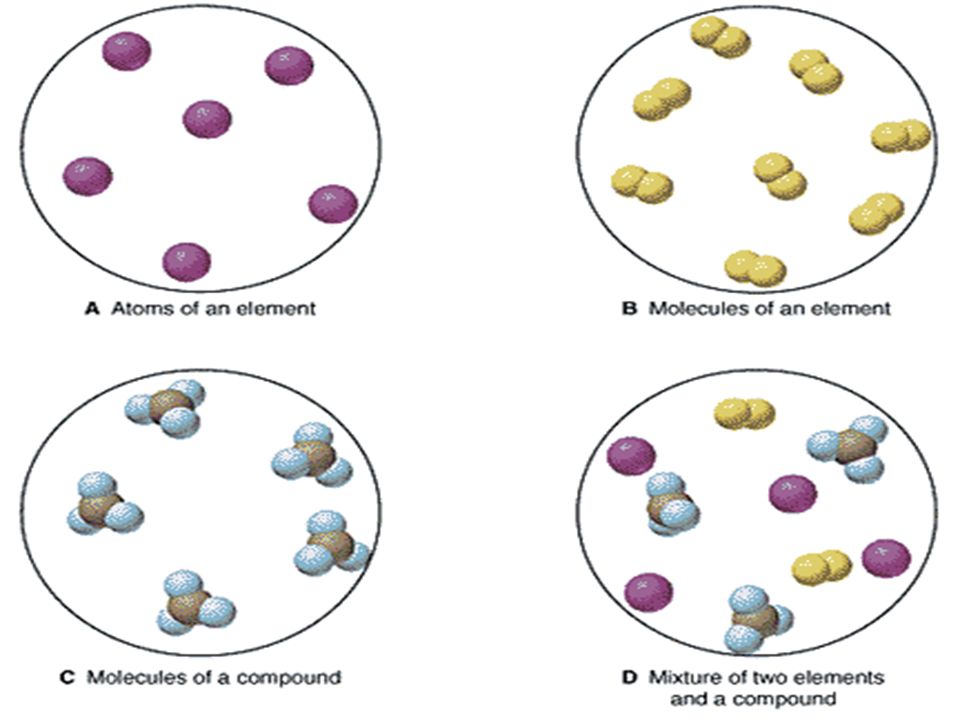
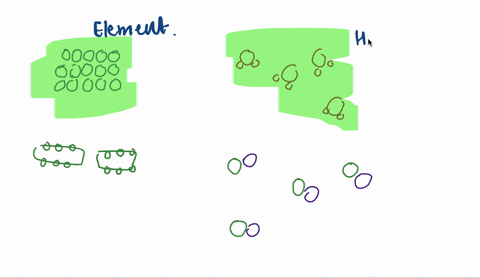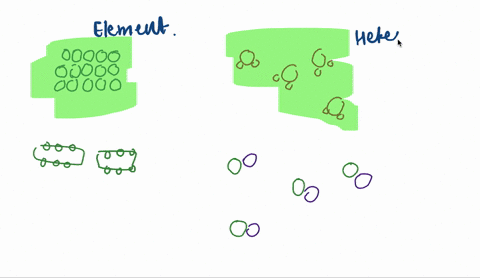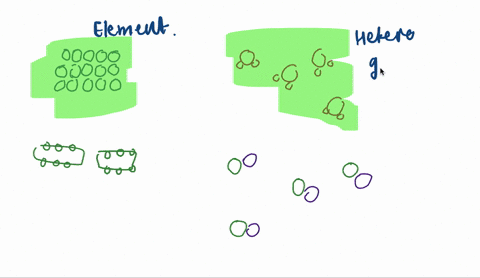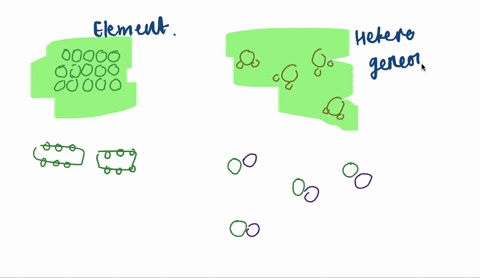
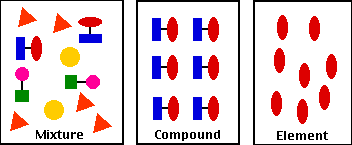
A heterogeneous mixture is a mixture with a nonuniform composition The composition varies from one region to another with at least two phases that remain separate from each other, with clearly identifiable properties If you examine a sample of a heterogeneous mixture, you can see the separate componentsRepresents an item composed of an element, compound, or mixture Review An element contains just one type of atom A compound contains two or more different atoms joined together A mixture contains two or more different substances that are only physically joined together, not chemically A mixture can contain both elements and compounds Two elements are liquid at room temperature — mercury and bromine Elements, gallium and caesium become liquid at a temperature slightly above room temperature (303 k) Differences between homogeneous and heterogeneous mixtures;




Mixtures And Their Separations




Elements Compounds And Mixtures Worksheet Chemistryrack
Mixtures in two or more phases are heterogeneous mixtures Examples include ice cubes in a drink, sand and water, and salt and oil Is paper heterogeneous or homogeneous?Mixtures Compounds Elements or compounds just mix together to form a mixture and no new compound is formed The main characteristic of a heterogeneous mixture is its lack of uniformity, that is, that it looks the same or that its properties do not vary where it is looked at or analyzed By having more than two distinguishable phases or components, according to the observation scale, uniformity is broken



1




Elements Componds Z 0001 Pdf
A heterogeneous mixture is often composed of different solids, or of matter existing in two or more different phases Phases are states of matter; By definition, a pure substance or a homogeneous mixture consists of a single phase A heterogeneous mixture consists of two or more phases When oil and water are combined, they do not mix evenly, but instead form two separate layers This phase would be homogeneousA heterogeneous mixture is composed of two or more phases When oil and water are combined, they do not mix evenly, but instead form two separate layers Each of the levels is called a phase Example (PageDex {1}) identify each substance as a compound, an element, a heterogeneous mixture or a homogeneous mixture (solution)



Homogeneous Vs Heterogeneous Mixtures A Comparison Expii



Elements Compounds Mixtures Matter Mixtures Multiple Substances Pure
Samar Arzoo Answered 2d 8h ago (a) Mixtures are substances that contain two or more different kinds of particles or substances mixed together by physical methods Examples Air, brick (b) (i) Mixtures in which the constituting substances are mixed together completely and are indistinguishable from one another are called homogeneous mixtures A mixture is the physical combination of two or more substances in which the identities are retained and are mixed in the form of solutions, suspensions and colloids 2 3 Mixtures are one product of mechanically blending or mixing chemical substances such as elements and compounds , without chemical bonding or other chemical change, so thatE) heating a mixture of two solids to fuse them together Answer D 16) Two or more substances in variable proportions, where the composition is variable throughout are A) a solution B) a homogeneous mixture C) a compound D) an amorphous solid E) a heterogeneous mixture Answer E 17) A physical change A) occurs when iron rusts



A Heterogeneous Mixture Composed Of The Two Elements Chegg Com




If Each Geometric Shape Represents A Different Type Of Atom And Atoms That Are Touching Have Brainly Com
6 rows A heterogeneous mixture is a type of mixture that allows the components to be seen as two or Yes, vinegar is a mixture that contains water and acetic acid (CH3COOH) The ratio of water and acetic acid is not definite So, it's definitely a mixture In science, a mixture is a matter that consists of two or more elements or compounds that combine together without aBy definition, a pure substance or a homogeneous mixture consists of a single phase A heterogeneous mixture consists of two or more phases When oil and water are combined, they do not mix evenly, but instead form two separate layers Each of the layers is called a phase



1



Www Gardencity K12 Ny Us Cms Lib Ny Centricity Domain 586 Elements compounds and particle diagrams 18 Pdf
Immiscible RRB ALP 10th Aug 18 Shift2 2 An alloy is an example of Solid solution Colloidal solution Emulsion Heterogeneous Heterogeneous Mixtures Mixtures which possess different properties and compositions in various parts of the mixture The chapter Elements, Compounds and Mixtures discusses the abovementioned topics in detail Once you finish studying the Chapter, try solving the Selina Questions and compare your answers with that provided by the BYJUâNot necessarily If the homogenization is done properly (because it is required) then the material could be nearly homogeneous or completely homogeneous in most of its parts See for example the following microstructures* From (a) to (f) you can




Pure Substances And Mixtures 1 Pure And Mixed



Homogeneous And Heterogeneous Mixtures Geeksforgeeks
A heterogeneous mixture is simply any mixture that is not uniform in composition it's a nonuniform mixture The constituent of this type of mixture are not evenly mixed throughout Using various means, the parts in the mixture can easily be separated from one another eg a tea leaves in a boiled tea, dusty air, a mixture of sand/oil in waterElement and Compound are examples of pure substances Mixtures are made up of two or more substances which are the constituents of mixture The constituents of mixture can be in any ratio Mixtures can be divided into 1 Homogeneous mixtures 2 Heterogeneous mixtures Homogeneous mixtures have uniform composition For instance, sugar inAn alloy is a material composed of two or more metals or a metal and non metal It may be a solid solution of the elements of single phase or a mixture of two or more metallic phases Solid alloys give a single solid phase microstructure Alloys are used where their properties are superior to those of pure component elements



Sxxv4y Ycx9hem




Which Best Represents A Gaseous Compound A Chegg Com
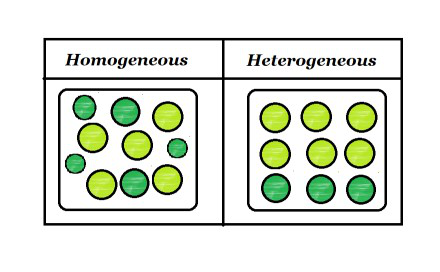
Homogeneous Vs Heterogeneous Mixture In chemistry, we can have two types of mixtures homogeneous mixtures and heterogeneous mixtures Homogeneous mixture Blended so thoroughly, it looks like one substance – Uniform composition Heterogeneous mixture Not thoroughly blended, so you can see and pick out an individual part of the mixture A mixture of two immiscible liquids is also a heterogeneous mixture, like a mixture of water and oil is also a heterogeneous mixture because when they both will be mixed together, the oil will start floating on the top surface, and they will not get mixed with each other forming two clear phases in the mixtureColloids and Suspensions When you drink a glass of whole or lowfat milk, you are drinking a mixture of water, fats, proteins, and other substances Milk is a colloid A colloid is a heterogeneous mixture that, like a solution, never settles One way to tell a colloid from a solution is show in the right figure Milk particles scatter light The scattering of light by particles In a mixture is called



1




Chapter 1 Review
In chemistry, a physical combination of two or more pure substances (ie, elements or compounds) A mixture is distinguished from a compound, which is formed by the chemical combination of two or more pure substances in a fixed, definite proportion Heterogeneous Mixtures A heterogeneous mixture is a mixture of two or more chemical substances (elements or compounds), where the different components can be visually distinguished and easily separated by physical meansA) It is possible to have heterogeneous mixtures in which all the components are liquids B) A homogeneous mixture contains visibly different parts, or phases C) All of the phases in a heterogeneous mixture must be in the same state D) No two phases in a heterogeneous mixture can be in the same state



Selina Icse Solutions For Class 9 Chemistry Elements Compounds And Mixtures A Plus Topper



Www Rcsdk12 Org Cms Lib04 Ny Centricity Domain 5551 Answer key for midterm packet Pdf
A mixture in which different materials can be easily distinguished heterogeneous mixture homogeneous mixture of particles so small they cannot be seen and will never settle to the bottom of their container solution substance in which two or more elements are combined in a fixed proportion compound ELEMENTS, COMPOUNDS & MIXTURE ,HETEROGENEOUS & HOMOGENOUS MIXTURE, COLLOIDS AND SUSPENSIONS RRB NTPC 12 April 16 Shift 3 1 When two liquids do not mix with each other to form a solution, what is it called ? Heterogeneous mixtures are made up of two or more totally different substances Homogeneous Mixture vs Heterogeneous Mixture A mixture is the combination of two or more pure substance in such a way that they are not chemically united The each pure substance getting into make the mixture have an influence on the mixture as it shows on the



3 4 Classifying Matter According To Its Composition Chemistry Libretexts




Matter Mixtures Pure Substances Elements Compounds Heterogenous Ppt Video Online Download
A heterogeneous mixture has a nonuniform composition, meaning that if several samples are taken, each would contain either completely different chemicals or the same chemicals, but in different ratios A chocolate chip cookie is a heterogeneous mixture, because it contains many different chemicals that form the cookie dough and the chocolate chips, which can be separated byThere are three wellknown phases gases, liquids, and solids A heterogeneous mixture is a combination of two or more substances, creating a new substance that has nonuniform composition Within the new substance, the concentration of the constituent substances will vary between regions of the substance, so it is possible for one of the chemicals to be more prominent towards the top of a heterogeneous mixture than at the




Difference Between Mixtures And Compounds In Tabular Form




2 3 Pure Substances Classification Of Matter Siyavula



1




2 Elements P3 Sukwon Sci




Mixture Homogeneous And Heterogeneous Mixtures Ck 12 Foundation



Lesson Categories Of Chemicals And Mixtures



Difference Between Homogeneous Heterogeneous Mixtures Biomadam




What Are The Types Of Pure Substances Compounds Elements Videos




Oneclass Classify Each Example As An Element A Compound A Heterogeneous Mixture Or A Homogeneous




Homogeneous Vs Heterogeneous Mixtures Difference And Comparison Diffen




Lesson Explainer Mixtures Nagwa




Chemistry The Central Science Chapter 1 Section 2




Unit 5 Pure Substances Mixtures Ppt Download




1 Which Of The Below Figures Represents A Gaseous Chegg Com




The A Eo 0 On A Through E Could Best Be Labeled Chegg Com



Www Dimanregional Org Site Handlers Filedownload Ashx Moduleinstanceid 39 Dataid 2719 Filename Cycle 1 chem 1 lesson 5 Pdf




Homogeneous Mixture Examples In Nature
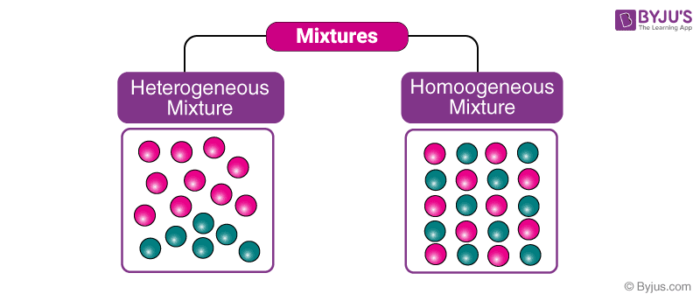



Heterogeneous And Homogeneous Mixture Differences Videos Examples




Lesson Explainer Mixtures Nagwa
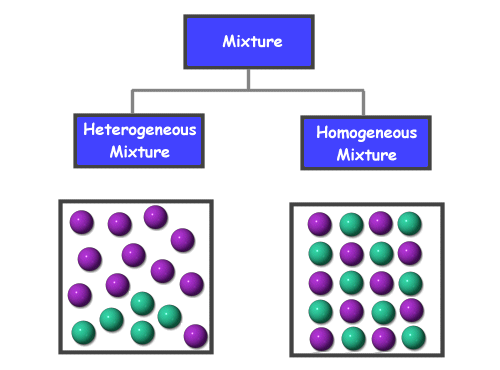



Chemistry For Kids Chemical Mixtures




Pure Substances And Mixtures Classification Of Matter Youtube
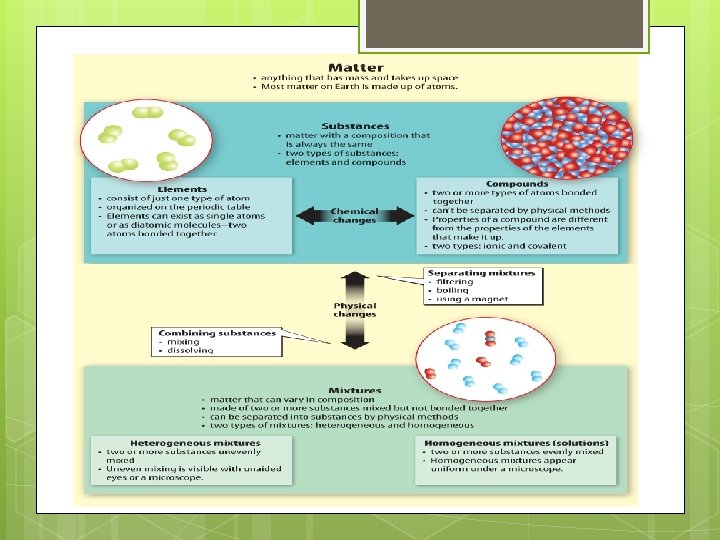



Mixtures Solubility And Acidbase Solutions Chapter 9 Chapter




Elements Compounds And Mixtures Activity Pdf Mixture Chemical Substances




Examples Of Homogeneous Mixtures Solid Liquid And Gas
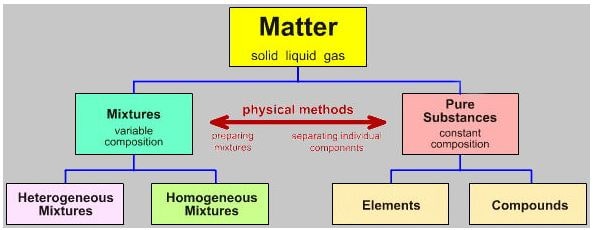



Difference Between Pure Substance And Mixture Guidance Corner



An Introduction To Chemistry Chapter 1 Hein And
/definition-of-heterogeneous-mixture-and-examples-605206_final23-ecfa4da6517640429448462eae1f09f7.png)



Definition Of Heterogeneous Mixture With Examples



Heterogeneous Mixture High Res Stock Images Shutterstock




Chapter 1 Section 2




Element Compound And Mixture Powerpoint Slides




Classification Of Matter Elements Compounds Mixtures Introductory Chemistry




What Is Matter Ck 12 Foundation



Pure Substances And Mixtures Neds Declassified



Elements Compounds And Mixtures Warm Up 9 25 1 Draw A Bohr Model For Boron 2 How Many Valence Electrons Does It Have Have 3 Do You Think It Is Ppt Download
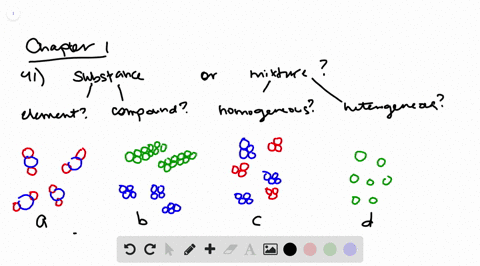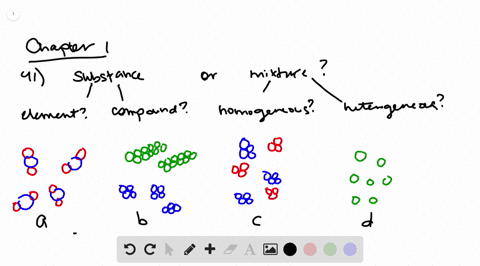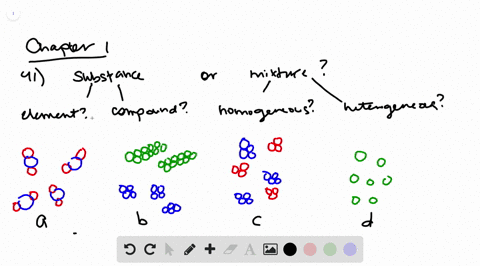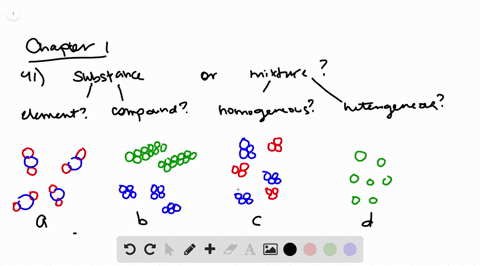



Solved Determine Whether Each Molecular Diagram Represents A Pure Substance Or A Mixture If It Represents A Pure Subst
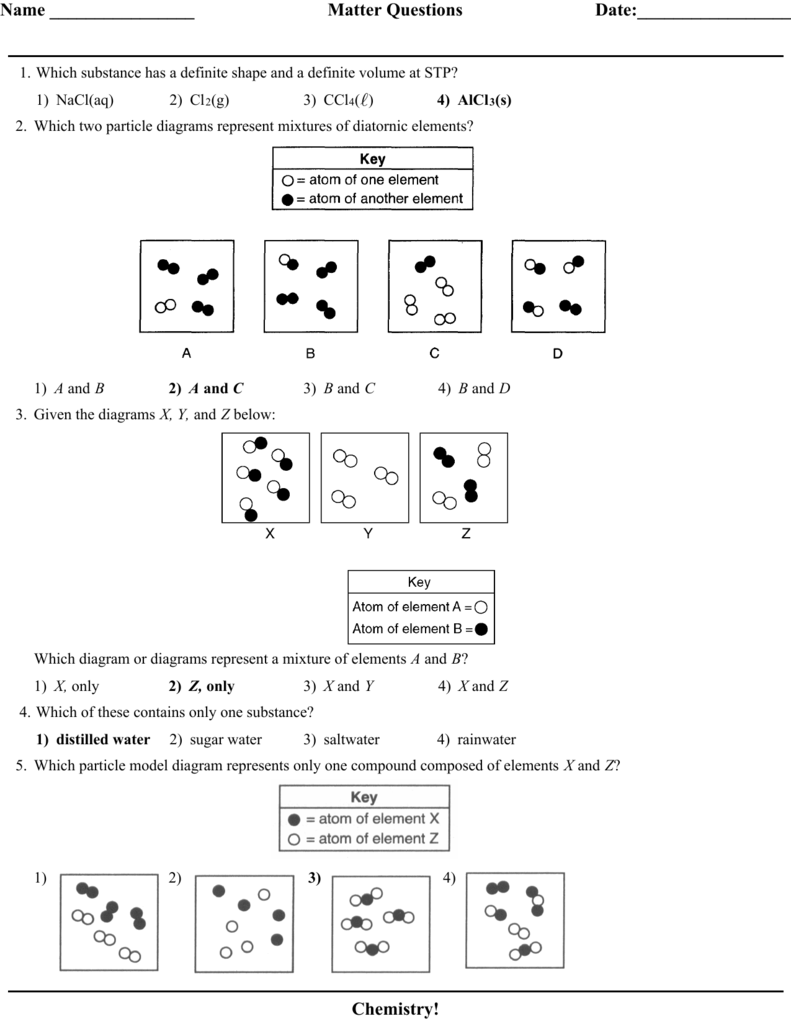



Matter Practice Quiz Answers




15 Pure Substance And Mixtures Ideas Teaching Science Chemistry Pure Products




What Do You Need To Know About Heterogeneous And Homogeneous Mixtures



Http Www B G K12 Ky Us Userfiles 1134 Classes Mixtures packet 13 14 Pdf




Elements Compounds Mixtures Worksheet Docest




Elements Compounds And Mixtures Diagram Quizlet




A Bit Of Explanation For The Second Picture Would Chegg Com




Test Ch 18 With Answers Saint Joseph High School




Types Of Matter How To Distinguish Elements Compounds Heterogeneous And Homogeneous Mixtures Youtube




Mixture Wikiwand




U2cc Particle Diagrams Youtube



Www Riohondoisd Net Site Handlers Filedownload Ashx Moduleinstanceid 1453 Dataid 4714 Filename 8th grade science brainpop Compounds And Mixtures Quiz a aguirre garcia Pdf




Sci C Un3clssfctnmttr



Unit 3 Pure Substances And Mixtures Ppt Video Online Download




10 Examples Of Mixtures




Elements Compounds Mixtures Review Diagram Quizlet
(22).jpg)



Can You Differentiate Between Various Chemical Substances Proprofs Quiz



Nanopdf Com Download File 7859 Pdf




Pure Substances Homogeneous Mixtures Solutions And Heterogeneous Mixtures Chapter 1 Part 2 Youtube



Www Redlandsusd Net Site Handlers Filedownload Ashx Moduleinstanceid Dataid Filename Exercise 1 10 classifying matter answers Pdf




Dublin Schools Lesson Particle Diagrams How Is Matter Represented In The Submicroscopic Domain
(120).jpg)



Chemistry Mixtures Elements Compounds Quiz Proprofs Quiz



Mixtures And Solutions Cpd Rsc Education



Is Matter Around Us Pure Practically Study Material




Solved Determine Whether Each Molecular Diagram Represents A Pure Substance Or A Mixture If It Represents A Pure Subst



Elements Compounds And Mixtures Sas




Element Mixture Compound Activity Teachengineering




Solved Sketch A Magnified View Showing Atoms Molecules Of Each Of The Following And Explain A A Heterogeneous Mixtu



Solved Sketch A Magnified View Showing Atoms Molecules Of Each Of The Following And Explain A A Heterogeneous Mixtu



3 Elements Compounds Mixtures




Heterogeneous Mixture High Res Stock Images Shutterstock



Mixtures And Compounds



Science Storyboard Par Tjskole




Scientific Milkshake Compunds Mixtures And Milkshakes




11 The Substance Represented In The Picture Is Best Chegg Com




Nia S Stuff Chemistry Homework 4



Ncert Class 9 Science Lab Manual Mixture And Compound Cbse Tuts
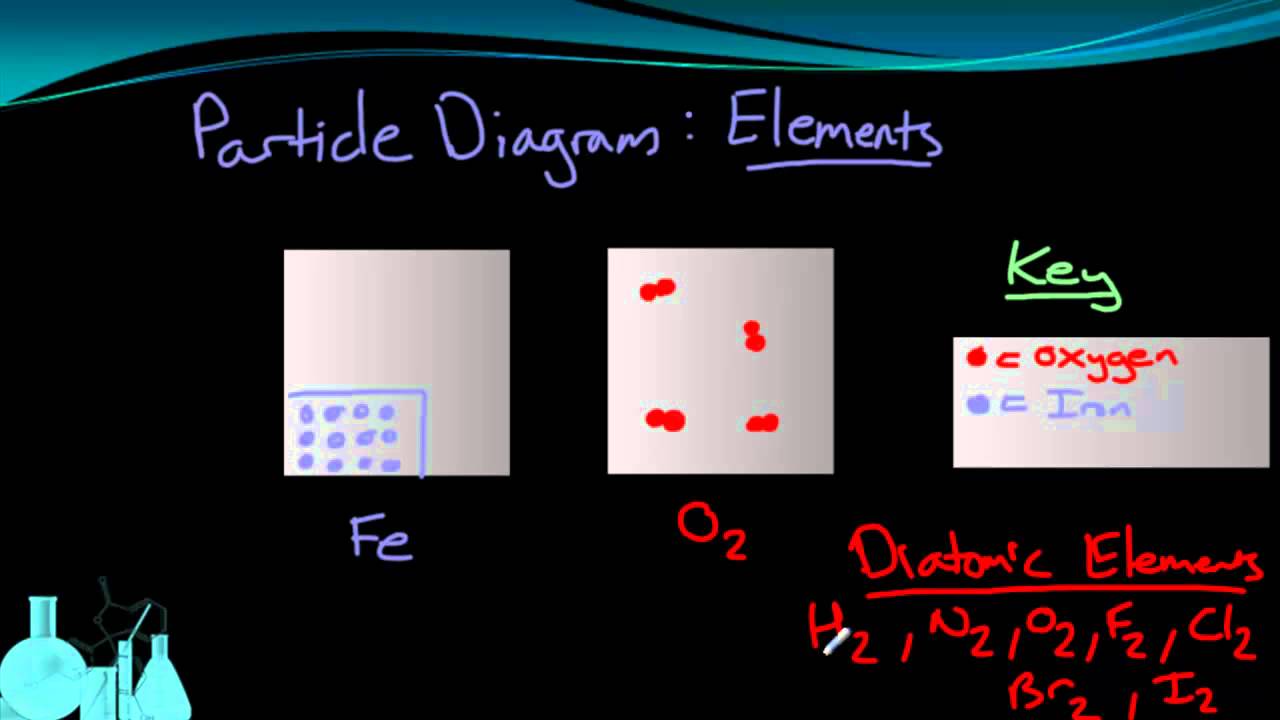



Classification Of Matter Elements Compounds Mixtures Introductory Chemistry




Mixtures A Mixture Is A Form Of Matter That Is Composed Of Two Or More Elements Two Or More Compounds Or Of Elements And Compounds Ppt Download



Mi Schoolwires Net Cms Lib05 Mi Centricity Domain 470 Intro hw packet key Pdf




Assignment Matter And Measurement Chemistry For Majors Atoms First



Is The Mixture Of Two Inert Gases An Example Of A Homogeneous Or Heterogeneous Mixture Quora



Sci8u1l2




10 Heterogeneous And Homogeneous Mixtures




Heterogeneous Mixture And Homogeneous Mixture Youtube




Homogeneous Vs Heterogeneous Mixtures A Comparison Expii




Elements Compounds And Mixtures Worksheet Answers




Lesson Explainer Mixtures Nagwa
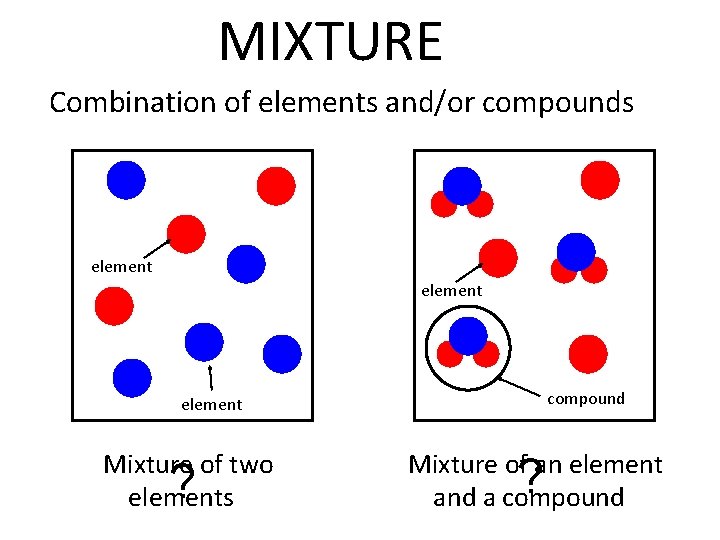



Matter Mixtures Mixtures Many Substances In Nature Are
コメント
コメントを投稿